Energy and Place
My contributions to the Joint Scientific Statement:
How Nuclear Fission Works
Energy is stored in the nuclei of atoms. Fission is when those nuclei are split and release lots of energy. It happens as such:
1. A neutron strikes the nucleus of a heavy and potentially unstable isotope.
2. The nucleus becomes unstable.
3. The nucleus vibrates and splits. The split occurring is fission.
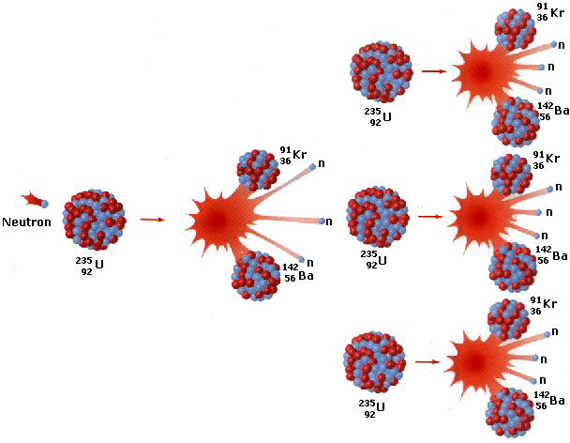
Since a fast neutron can’t be captured, neutrons are slowed down through moderation in order to increase their capture probability in fission reactors. A single fission event can yield over 200 million times the energy of the neutron which triggered it! Fission produces two or three neutrons, two new, lighter weight atoms called fission products, and energy (mostly as heat). The energy that is released is that which was holding the hadrons (neutrons and protons) together. Chain reactions are when fission occurs, and the neutrons released from that hit more atoms, releasing more neutrons, which hit more atoms. The results in lots of energy! For example, in U-235, a slow neutron is captured by the atom’s nucleus, turning it into U-236 and making it unstable. The nucleus splits, producing Kr-92 and Ba-141 and sending more neutrons off that can start a chain reaction. This can be seen in the visual below.
How Nuclear Fission Works
Energy is stored in the nuclei of atoms. Fission is when those nuclei are split and release lots of energy. It happens as such:
1. A neutron strikes the nucleus of a heavy and potentially unstable isotope.
2. The nucleus becomes unstable.
3. The nucleus vibrates and splits. The split occurring is fission.
Since a fast neutron can’t be captured, neutrons are slowed down through moderation in order to increase their capture probability in fission reactors. A single fission event can yield over 200 million times the energy of the neutron which triggered it! Fission produces two or three neutrons, two new, lighter weight atoms called fission products, and energy (mostly as heat). The energy that is released is that which was holding the hadrons (neutrons and protons) together. Chain reactions are when fission occurs, and the neutrons released from that hit more atoms, releasing more neutrons, which hit more atoms. The results in lots of energy! For example, in U-235, a slow neutron is captured by the atom’s nucleus, turning it into U-236 and making it unstable. The nucleus splits, producing Kr-92 and Ba-141 and sending more neutrons off that can start a chain reaction. This can be seen in the visual below.
The Greenhouse Effect and Its Relation to Global Climate Change
Without the greenhouse effect, temperatures on Earth would not be livable. When the sun’s radiation reaches our atmosphere, some is redirected back into space, while some reach through and heat up the surface of the Earth. The heat from the Earth’s surface is then radiated outward and caught in the greenhouse gases contained in our atmosphere. This keeps heat from escaping into space. While this is vital to our survival and life as a whole, it is also why the Earth is heating up so rapidly. The burning of fossil fuels and other activities intensifies the warming effect by releasing additional greenhouse gases into the atmosphere (such as carbon dioxide and methane). Greenhouse gases come from various sources and all have different heat trapping capabilities.
Greenhouse Gases and Their Structure
Greenhouses gases are gases that trap heat in the atmosphere. The greenhouse gases found in our atmosphere are carbon dioxide, methane, nitrous oxide, and fluorinated gases. Despite being a relatively low greenhouse gas, so much CO2 has been emitted since pre-industrial times that it has had the largest impact on the enhanced greenhouse effect. In 2012 it accounted for 82% of all greenhouse emissions! It is produced primarily by fossil fuels that are burned through the use of vehicles, power plants, and industry, and emissions continue to increase every year. Methane only makes up less than two of every million molecules in the air and stays in our atmosphere for only ten years. Despite this, methane traps 20 to 25 times as much energy in that decade as carbon dioxide does in a century. Nitrous oxide accounts for only about 300 in every billion molecules in the air, but is 300 times more powerful than CO2 at enhancing the greenhouse effect and stays in our atmosphere for around 100 years. Carbon dioxide (CO2), water vapor (H2O), methane (CH4, nitrous oxide (N2O) are all molecules composed of more than two loosely bound component atoms. These molecules can absorb radiation because they are able to vibrate with the absorption of heat. The two main components of the atmosphere (N2 and O2) are too tightly bound to vibrate and as a result do not absorb heat. When they absorb radiation, they begin to vibrate. Eventually, it will re-emit the radiation that it absorbed. That radiation will most likely be absorbed by another greenhouse gas in the atmosphere. The absorption-emission-absorption cycle effectively keeps the atmosphere warm.
Debate contributions:
Debate Opening Statement
One of the biggest risks of nuclear energy is the potential for a nuclear disaster to occur. Since 1952, there have been at least 33 accidents and incidents involving nuclear power stations, 20 of which resulted in fatalities. The consequences of these nuclear disasters are so much worst than those of other energy sources. Notable nuclear disasters include the Kyshtym disaster, Fukushima disaster, and the Chernobyl disaster.
When discussing whether or not we should go forward with nuclear power, the Chernobyl disaster cannot be overlooked. The Chernobyl disaster of 1986 was the worst nuclear disaster to ever occur, releasing radioactive material that spread over parts of the Soviet Union and Europe, the radioactivity of the nuclear disaster being more than 400 times potent than that of the atomic bomb dropped on Hiroshima. While only 30 people died in the immediate aftermath (due to the explosion or acute radiation syndrome), those living in the contaminated zones have seen increases in both thyroid cancer and birth defects.
The past has shown us that nuclear energy leads to problems such as these, so we must ask ourselves, with so many other energy sources at hand, is nuclear energy really what we want to move forward with?
Debate Closing Statement
Before you decide on which side of the argument is declared winner, I'd like you to observe these pictures. These are all children, born years after the Chernobyl disaster. Figures released by UNICEF in 2010 showed that more than 20% of adolescent children in Belarus suffered from disabilities and chronic illness. Belarus absorbed 70% of Chernobyl's fallout. So, please, everyone, I urge you. If you won’t vote against nuclear power because of the damages of mining uranium or the cost of building nuclear power plants, then please, do it for the children.
Without the greenhouse effect, temperatures on Earth would not be livable. When the sun’s radiation reaches our atmosphere, some is redirected back into space, while some reach through and heat up the surface of the Earth. The heat from the Earth’s surface is then radiated outward and caught in the greenhouse gases contained in our atmosphere. This keeps heat from escaping into space. While this is vital to our survival and life as a whole, it is also why the Earth is heating up so rapidly. The burning of fossil fuels and other activities intensifies the warming effect by releasing additional greenhouse gases into the atmosphere (such as carbon dioxide and methane). Greenhouse gases come from various sources and all have different heat trapping capabilities.
Greenhouse Gases and Their Structure
Greenhouses gases are gases that trap heat in the atmosphere. The greenhouse gases found in our atmosphere are carbon dioxide, methane, nitrous oxide, and fluorinated gases. Despite being a relatively low greenhouse gas, so much CO2 has been emitted since pre-industrial times that it has had the largest impact on the enhanced greenhouse effect. In 2012 it accounted for 82% of all greenhouse emissions! It is produced primarily by fossil fuels that are burned through the use of vehicles, power plants, and industry, and emissions continue to increase every year. Methane only makes up less than two of every million molecules in the air and stays in our atmosphere for only ten years. Despite this, methane traps 20 to 25 times as much energy in that decade as carbon dioxide does in a century. Nitrous oxide accounts for only about 300 in every billion molecules in the air, but is 300 times more powerful than CO2 at enhancing the greenhouse effect and stays in our atmosphere for around 100 years. Carbon dioxide (CO2), water vapor (H2O), methane (CH4, nitrous oxide (N2O) are all molecules composed of more than two loosely bound component atoms. These molecules can absorb radiation because they are able to vibrate with the absorption of heat. The two main components of the atmosphere (N2 and O2) are too tightly bound to vibrate and as a result do not absorb heat. When they absorb radiation, they begin to vibrate. Eventually, it will re-emit the radiation that it absorbed. That radiation will most likely be absorbed by another greenhouse gas in the atmosphere. The absorption-emission-absorption cycle effectively keeps the atmosphere warm.
Debate contributions:
Debate Opening Statement
One of the biggest risks of nuclear energy is the potential for a nuclear disaster to occur. Since 1952, there have been at least 33 accidents and incidents involving nuclear power stations, 20 of which resulted in fatalities. The consequences of these nuclear disasters are so much worst than those of other energy sources. Notable nuclear disasters include the Kyshtym disaster, Fukushima disaster, and the Chernobyl disaster.
When discussing whether or not we should go forward with nuclear power, the Chernobyl disaster cannot be overlooked. The Chernobyl disaster of 1986 was the worst nuclear disaster to ever occur, releasing radioactive material that spread over parts of the Soviet Union and Europe, the radioactivity of the nuclear disaster being more than 400 times potent than that of the atomic bomb dropped on Hiroshima. While only 30 people died in the immediate aftermath (due to the explosion or acute radiation syndrome), those living in the contaminated zones have seen increases in both thyroid cancer and birth defects.
The past has shown us that nuclear energy leads to problems such as these, so we must ask ourselves, with so many other energy sources at hand, is nuclear energy really what we want to move forward with?
Debate Closing Statement
Before you decide on which side of the argument is declared winner, I'd like you to observe these pictures. These are all children, born years after the Chernobyl disaster. Figures released by UNICEF in 2010 showed that more than 20% of adolescent children in Belarus suffered from disabilities and chronic illness. Belarus absorbed 70% of Chernobyl's fallout. So, please, everyone, I urge you. If you won’t vote against nuclear power because of the damages of mining uranium or the cost of building nuclear power plants, then please, do it for the children.
Reflection
This project had us get into groups of seven, six researchers and a moderator. The seven of us were required to research nuclear energy and construct a Joint Scientific Statement. After, the six researchers were divided into two groups, one for and one against nuclear energy. I was arguing against nuclear energy.
This opposed my actual viewpoint; I am actually for nuclear energy. I did not find this challenging, however. I don't find it difficult to argue from perspectives other than mine. While my original position was not unchanged, learning of nuclear disasters and their lost-lasting effects did make me question nuclear energy a bit. Fortunately, I believe we have the means available to prevent anything major (such as Chernobyl) from happening again.
I found the best arguments against nuclear energy to be the dangers of running a nuclear power plant and the risk of disaster and the costs of building a plant. The best arguments for nuclear energy were the low amounts of greenhouse gases it produces, and the relative cheapness to gas after the plant is built.
I would like to look more into the cost of different energy sources, as I did not delve very deep into that for my debate.
My environmental ethic of conservationism is a huge reason why I support nuclear energy. I believe in sustaining the Earth for future use of its resources. I wish I would have done better during my debate, as I did not really give my group much support. I was extremely exhausted during my debate and a little underprepared. I feel like I did bring up some good points with nuclear disasters and that I did well to show them the effects Chernobyl had on some children. Ultimately, I had strong opening and closing statements but struggled in the open debate.
You can see the Humanities side to this project here.
This opposed my actual viewpoint; I am actually for nuclear energy. I did not find this challenging, however. I don't find it difficult to argue from perspectives other than mine. While my original position was not unchanged, learning of nuclear disasters and their lost-lasting effects did make me question nuclear energy a bit. Fortunately, I believe we have the means available to prevent anything major (such as Chernobyl) from happening again.
I found the best arguments against nuclear energy to be the dangers of running a nuclear power plant and the risk of disaster and the costs of building a plant. The best arguments for nuclear energy were the low amounts of greenhouse gases it produces, and the relative cheapness to gas after the plant is built.
I would like to look more into the cost of different energy sources, as I did not delve very deep into that for my debate.
My environmental ethic of conservationism is a huge reason why I support nuclear energy. I believe in sustaining the Earth for future use of its resources. I wish I would have done better during my debate, as I did not really give my group much support. I was extremely exhausted during my debate and a little underprepared. I feel like I did bring up some good points with nuclear disasters and that I did well to show them the effects Chernobyl had on some children. Ultimately, I had strong opening and closing statements but struggled in the open debate.
You can see the Humanities side to this project here.
Materials Unit Project
Lithium-Sulfur Batteries Are The Future
Travis Carlson
As time wears on, the need for more energy efficient ways of powering our vehicles and devices is increasing. Demand for electric cars that can match up to cars with combustion engines is also becoming more apparent. The lithium-ion batteries currently found in electric cars are able to get the job done, but they only allows us to go so far. I am proposing a new battery that is being researched. This battery, instead of being made of lithium-ion, is made of lithium sulfur.
Demand for electric cars that can match up to cars with combustion engines has brought scientists to research more effective battery than lithium-ion batteries. Lithium-sulfur batteries offer a cheaper, more dense battery that have more than twice the specific energy of lithium-ion batteries, while being capable of lasting more than 1,500 charge cycles with minimal decay of the battery's capacity. This is the highest amount of energy a lithium-sulfur battery has held to date, and thus, provides a much longer cycle life than lithium-ion batteries.
Lithium-sulfur batteries are desirable over lithium-ion batteries because sulfur is non-toxic, inexpensive, and safe. Overheating, catching fire, and other safety problems have caused trouble for the current lithium-ion batteries. Lithium-sulfur batteries are less prone to these problems, while at the same time being a more cost-effective battery. A novel ionic liquid based electrolyte was developed by the team of scientists working on the lithium-sulfur battery. This allows the battery to be incredibly safe (ionic liquids are non-volatile and non-flammable) while helping the battery operate at a high rate, increasing the speed at which the battery can be charged up, and the power it can deliver during discharge (Chen).
Scientists working on the lithium-sulfur batteries suggest that a non-stop trip in an eco-friendly vehicle between London and Paris could be possible under a single charge. According to thechargingpoint.com, the average charge of a lithium-ion battery only allows cars powered by it to go around one-hundred miles before needing a recharge. The lithium-sulfur battery will triple that, and bring us one step closer to clean fuel everywhere.
There are some concerns surrounding lithium-sulfur batteries, though. One is a problem with cell degradation. During discharge lithium polysulfides tend to dissolve from the cathode in the electrolytes and react with the lithium anode forming a barrier layer of Li2S (Chen). Another concern is that of the conversion reaction, which causes the volume of the sulfur electrode to swell and contract up to 76 percent during cell operation. This allows sulfur molecules to become electrically isolated from the current collector of the electrode.
Increasing the cell energy density, improving cell performance under extreme condition, and scale up to larger cells are the next steps in development for the Lithium-sulfur battery.
Reflection
How has the chemistry of materials shaped our past, present and how may it shape our future?
The chemistry of materials have shaped our past and present is so many ways. Most, if not all, of the materials we use can be looked at from a chemistry viewpoint. The use of chemistry has allowed us to create new materials as well, such as the batteries I listed above. Chemistry is pretty sweet if you ask me. Without the understanding of the chemistry of materials we have today, we wouldn't be where we are. I think it will just make our future better and better as we learn more.
How does the structure of matter on the atomic, molecular, microscopic and macroscopic levels determine a material’s properties?
The structure determines so many of a material's properties. Whether it is crystalline or amorphous, whether it is
Travis Carlson
As time wears on, the need for more energy efficient ways of powering our vehicles and devices is increasing. Demand for electric cars that can match up to cars with combustion engines is also becoming more apparent. The lithium-ion batteries currently found in electric cars are able to get the job done, but they only allows us to go so far. I am proposing a new battery that is being researched. This battery, instead of being made of lithium-ion, is made of lithium sulfur.
Demand for electric cars that can match up to cars with combustion engines has brought scientists to research more effective battery than lithium-ion batteries. Lithium-sulfur batteries offer a cheaper, more dense battery that have more than twice the specific energy of lithium-ion batteries, while being capable of lasting more than 1,500 charge cycles with minimal decay of the battery's capacity. This is the highest amount of energy a lithium-sulfur battery has held to date, and thus, provides a much longer cycle life than lithium-ion batteries.
Lithium-sulfur batteries are desirable over lithium-ion batteries because sulfur is non-toxic, inexpensive, and safe. Overheating, catching fire, and other safety problems have caused trouble for the current lithium-ion batteries. Lithium-sulfur batteries are less prone to these problems, while at the same time being a more cost-effective battery. A novel ionic liquid based electrolyte was developed by the team of scientists working on the lithium-sulfur battery. This allows the battery to be incredibly safe (ionic liquids are non-volatile and non-flammable) while helping the battery operate at a high rate, increasing the speed at which the battery can be charged up, and the power it can deliver during discharge (Chen).
Scientists working on the lithium-sulfur batteries suggest that a non-stop trip in an eco-friendly vehicle between London and Paris could be possible under a single charge. According to thechargingpoint.com, the average charge of a lithium-ion battery only allows cars powered by it to go around one-hundred miles before needing a recharge. The lithium-sulfur battery will triple that, and bring us one step closer to clean fuel everywhere.
There are some concerns surrounding lithium-sulfur batteries, though. One is a problem with cell degradation. During discharge lithium polysulfides tend to dissolve from the cathode in the electrolytes and react with the lithium anode forming a barrier layer of Li2S (Chen). Another concern is that of the conversion reaction, which causes the volume of the sulfur electrode to swell and contract up to 76 percent during cell operation. This allows sulfur molecules to become electrically isolated from the current collector of the electrode.
Increasing the cell energy density, improving cell performance under extreme condition, and scale up to larger cells are the next steps in development for the Lithium-sulfur battery.
Reflection
How has the chemistry of materials shaped our past, present and how may it shape our future?
The chemistry of materials have shaped our past and present is so many ways. Most, if not all, of the materials we use can be looked at from a chemistry viewpoint. The use of chemistry has allowed us to create new materials as well, such as the batteries I listed above. Chemistry is pretty sweet if you ask me. Without the understanding of the chemistry of materials we have today, we wouldn't be where we are. I think it will just make our future better and better as we learn more.
How does the structure of matter on the atomic, molecular, microscopic and macroscopic levels determine a material’s properties?
The structure determines so many of a material's properties. Whether it is crystalline or amorphous, whether it is